Moderna on X: "A Phase 2/3 trial for mRNA-1273.222 is currently underway. We have rapidly scaled manufacturing of mRNA-1273.222 to be ready, if authorized, to deliver doses in September. https://t.co/AQG4hfwNe6" / X
Moderna's BA.4/BA.5 Targeting Bivalent Booster, mRNA-1273.222, Meets Primary Endpoint of Superiority Against Omicron Variants Compared to Booster Dose of mRNA-1273 in Phase 2/3 Clinical Trial

Moderna on X: "In a Phase 2/3 trial in over 500 adults, mRNA-1273.222, induced significantly higher neutralizing antibody titers against BA.4/BA.5 compared to a booster dose of mRNA-1273. Read more: https://t.co/CPnxL03nZW https://t.co/ODihw6qtsQ" /
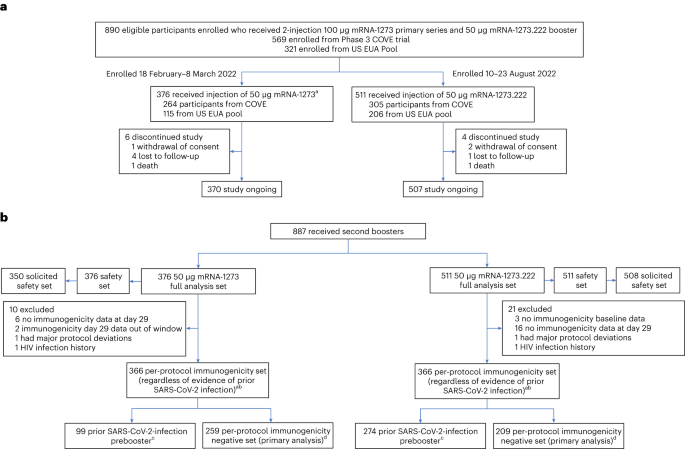
Original SARS-CoV-2 monovalent and Omicron BA.4/BA.5 bivalent COVID-19 mRNA vaccines: phase 2/3 trial interim results | Nature Medicine

Bivalent SARS-CoV-2 mRNA vaccines increase breadth of neutralization and protect against the BA.5 Omicron variant in mice | Nature Medicine